-

-

Blog
Rare disease trials don’t need better recruitment. They need awareness.
8 min
●April 3, 2026
SubjectWell Team -

-

Blog
How global standards of care data shape patient recruitment
6 min
●March 3, 2026
SubjectWell Team -

Blog
The future of global patient engagement starts with experience
4 min
●February 18, 2026
SubjectWell Team

A peer-reviewed, scientific approach to online recruitment in a depression study
- CRO, Site Network, Sites, Sponsors
- Digital Marketing, Study Connect, Study support
- Mental Health

In our Behind the Screens series we are spotlighting our Site Companions: the dedicated site engagement specialists, who power site support and clinical trial efficiency.
- Patient and Site Companions, Site Support

In this Behind the Screens spotlight, we feature Merusha Naidoo, a site team leader whose relationship-driven approach helps ease burden and boost performance across trial sites around the world.
- Patient and Site Companions, Site Support

SubjectWell CEO Fred Martin explores why symbolic support isn’t enough and how the steps the clinical trial industry needs to make to drive equity, representation, and better outcomes for LGBTQ+ individuals.
- CRO, Sponsors
- Patient Care & Trial Retention, Patient Support
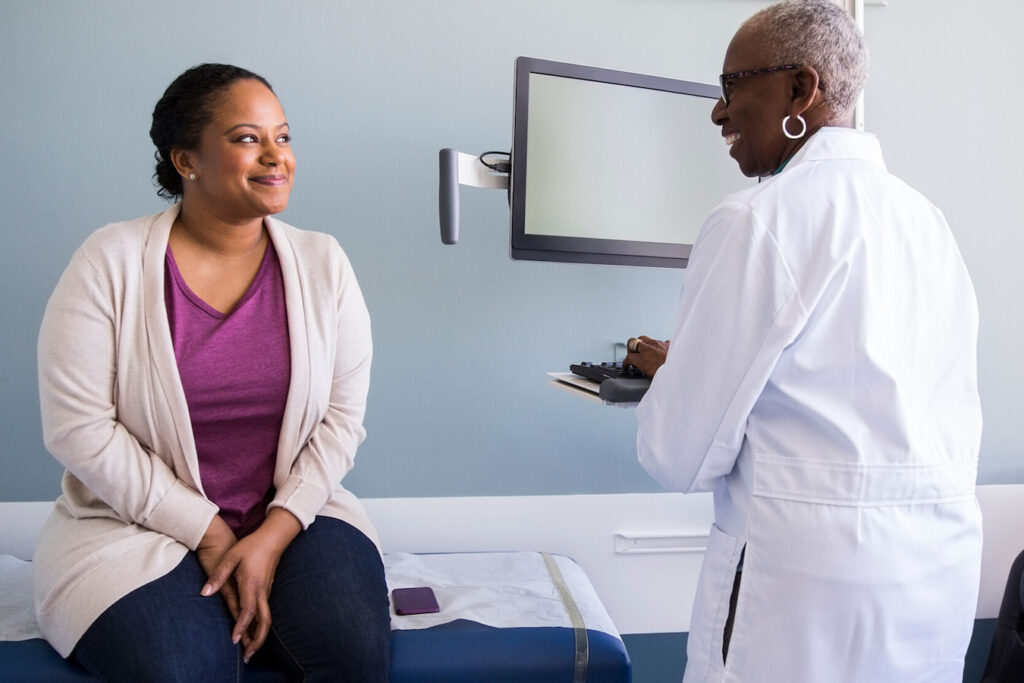
Learn how SubjectWell tackles key barriers in psychiatric trials—like stigma, documentation delays, and caregiver burden—with services that improve engagement, retention, and site efficiency.
- CRO, Site Network, Sites, Sponsors
- OneView with Instant Access Medical Records, Patient Care & Trial Retention, Patient Support, Secondary Screening, Site Support, Study support, Translations, Virtual Waiting Room
- Mental Health

Not every GLP-1 trial participant sees their condition in the same way. From diabetes to obesity and addiction, patient mindsets influence trial participation. Learn why tailored engagement strategies are essential for successful GLP-1 recruitment.
- CRO, Sponsors
- Full-Site Support, Patient and Site Companions, Patient Care & Trial Retention, Patient Companions, Patient Support, Secondary Screening, Site Support, Study support, Translations

Learn about a day in the life of a Patient Companion at SubjectWell and how they support patients throughout their clinical trial journey.
- CRO, Sites, Sponsors
- Patient Companions, Patient Support

Transform your clinical trials with patient-centric approaches and innovative recruitment strategies.
- CRO, Sites, Sponsors
- OneView with Instant Access Medical Records, Patient and Site Companions, Patient Care & Trial Retention, Patient Companions, Patient Panels & Surveys, Patient Support, Recruitment Simulation, Site Support, Study support

Learn about a day in the life of a Patient Companion at SubjectWell and how they support patients throughout their clinical trial journey.
- CRO, Sponsors
- Patient Companions, Patient Support
WHY SUBJECTWELL
It’s time to transform patient recruitment
-
300,000
patients added monthly to our network
47%
registered patients of color
100+
clinically trained patient companions
-
300,000
patients added monthly to our network
47%
registered patients of color
100+
clinically trained patient companions
-
300,000
patients added monthly to our network
47%
registered patients of color
100+
clinically trained patient companions
Their tailored approach helped us meet our recruitment target ahead of schedule. Highly recommend!
Name
Job Description
Accelerate your recruitment today
SubjectWell is here to help you reach, recruit, and retain the patients you need for a successful clinical trial. Schedule a demo to see how SubjectWell+ can support your clinical study’s patient recruitment goals.